Download Our Cryopreservation Equipment Overview Booklet
Moving into a New Era of Cryosafety
 As we progress toward Q2 of 2026, the cryopreservation industry is proactively moving away from the time of passive storage, where the primary goal was simply avoiding a tank failure, and toward a new, more complex, era of “Cryosafety 2.0”. This shifting mindset views the cryopreservation unit not just as a freezer, but as an active, digital participant in the clinical chain of custody.
As we progress toward Q2 of 2026, the cryopreservation industry is proactively moving away from the time of passive storage, where the primary goal was simply avoiding a tank failure, and toward a new, more complex, era of “Cryosafety 2.0”. This shifting mindset views the cryopreservation unit not just as a freezer, but as an active, digital participant in the clinical chain of custody.
What does "Cryosafety 2.0" mean?
The core philosophy of Cryosafety 2.0 is that identity and process failures are now as dangerous as mechanical failures. In the UK’s current regulatory climate, the safety of a sample is defined by three new pillars, i.e., active traceability, lid-open vulnerability awareness and audit-ready automation, so let’s look at why.
Active Traceability: In modern cryogenic storage environments, merely keeping a sample at the correct temperature (e.g., below -135°C) is no longer enough. Labs must now provide complete traceability of every interaction with that sample throughout its lifecycle, from receipt to retrieval. Advanced traceability systems, such as Bluechiip’s MEMS device, readers and software, integrate barcode or RFID identification with sample management software so that every individual tube or vial is uniquely tracked within the inventory, linked to donor data, storage location, project and experimental metadata. This digital chain of custody ensures that not only is the sample kept in optimal conditions, but also who accessed it, when, and where it was stored are logged and auditable too. This meets regulators’ expectations for confidentiality, integrity and availability of data and aligns with best-practice traceability standards used in clinical and biopharma biobanks.
Lid-Open Vulnerability Awareness: Traditional cryogenic freezers, particularly manual liquid nitrogen (LN₂) tanks and ultra-low-temperature units, suffer from a well-recognised physical limitation, when the lid or door is opened, warm air intrudes and causes transient warming of both target and neighbouring samples. Studies and industry reports show that even brief temperature excursions above the ideal cryogenic set-point can materially degrade cell viability, particularly for sensitive cell therapy products like CAR-T cells.
Thermal shock not only threatens sample integrity but also leads to operational inefficiencies as personnel must work quickly and often under suboptimal ergonomic conditions to reduce exposure times.
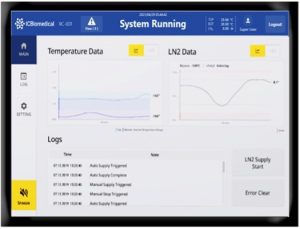
 Audit-Ready Automation: With regulatory bodies like the UK’s Human Fertilisation and Embryology Authority (HFEA), the Human Tissue Authority (HTA), the FDA and other global authorities increasing the frequency and digital depth of compliance audits, laboratories are under pressure to move away from manual logs and paper records entirely. The latest cryogenic storage solutions are integrating live audit trails, where every event affecting the freezer and its samples is automatically time-stamped and linked to a unique user ID.
Audit-Ready Automation: With regulatory bodies like the UK’s Human Fertilisation and Embryology Authority (HFEA), the Human Tissue Authority (HTA), the FDA and other global authorities increasing the frequency and digital depth of compliance audits, laboratories are under pressure to move away from manual logs and paper records entirely. The latest cryogenic storage solutions are integrating live audit trails, where every event affecting the freezer and its samples is automatically time-stamped and linked to a unique user ID.
How the latest technology helps combat these issues
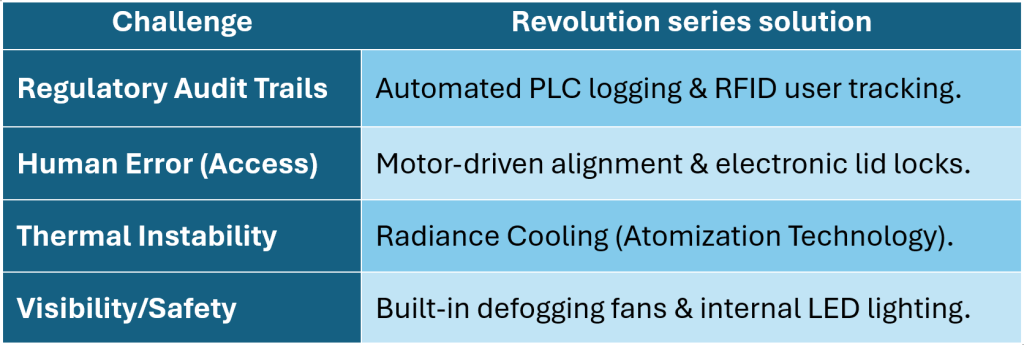
Biopharma Group offers* the latest innovations from IC Biomedical including the Revolution Series which is engineered precisely to address the challenges posed by a “Cryosafety 2.0” world. Here is how it solves the specific hurdles faced by UK labs today.
Industry Challenge vs. Revolution series solution
Solving the identity risk or the digital chain of custody
The problem with manual ‘double witnessing’ of sample handling is that it is prone to human error and is labour-intensive. Featuring an integrated RFID badge reader and a PLC-based touchscreen, the Revolution series tackles this challenge head-on as a technician cannot unlock the lid without a verified badge. The system then generates a full audit trail that records exactly which user accessed the unit and for how long, fulfilling the most stringent HTA/HFEA ‘witnessing’ requirements without a second staff member being present.
 Eliminating transient warming
Eliminating transient warming
Every time a large tank is opened, the vapour cloud escapes and the top-row samples are at risk of going above the critical glass transition temperature (circa -135°C), compromising more than just the sample being retrieved. Built-in extraction fan to clear fog when the lid is opened, allows quicker access to samples avoiding the heat spike that typically threatens sample viability. Together with the inbuilt LED light, the time spent with lid open to identify the required samples is kept to a minimum.
Reducing physical handling errors through automation
Manually rotating heavy, frozen carousels to find a specific rack is core source of time lost in searching for samples, which keeps the lid open longer and increases the risk of dropping or misidentifying samples. With the option of a motor-driven carousel with rack learning technology, the Revolution series addresses this common issue. The user selects a sample on the screen and the carousel rotates automatically to align that specific rack directly under the lid opening. This minimizes lid-open time, ensures the technician only interacts with the correct section of the tank and is working within the freezer for the shortest period.
Future proofing for ‘Green’ UK mandates
UK labs are under pressure to reduce Liquid Nitrogen (LN2) consumption due to rising supply chain costs and sustainability targets. The Revolution series has been developed to have some of the lowest LN2 consumption rates in the industry. Its design allows it to maintain a minimum temperature hold in the event of a total supply failure, providing a massive safety buffer for UK labs that may face logistics delays.
The evolution of cryogenic storage freezers to meet the demands of the new cryopreservation landscape, such as the advances led by IC Biomedical, are enabling laboratories and repositories to future proof their operations while ensuring compliance, safety and operational efficiency. With advanced controllers, remote monitoring and enhanced security features, laboratories can now manage and safeguard their samples more effectively than ever before.
To discover more, get in touch* with a specialist today.
*Available to the UK, Ireland & France
